VZV-VM10E-100UG VZV-VM10E-500UG
Scientist.com Supplier
Varicella-zoster virus (strain Oka vaccine) Envelope glycoprotein E Protein (VZV-VM10E)
KactusBio
Image
Image
Image
DESCRIPTION
Recombinant Varicella-zoster virus (strain Oka vaccine) Envelope glycoprotein E Protein is expressed from HEK293 with His tag at the C-Terminus.; It contains Ser31-Tyr538.
DETAILS
- Tag: C-His
- Form: Lyophilized
- Type: Recombinant Protein
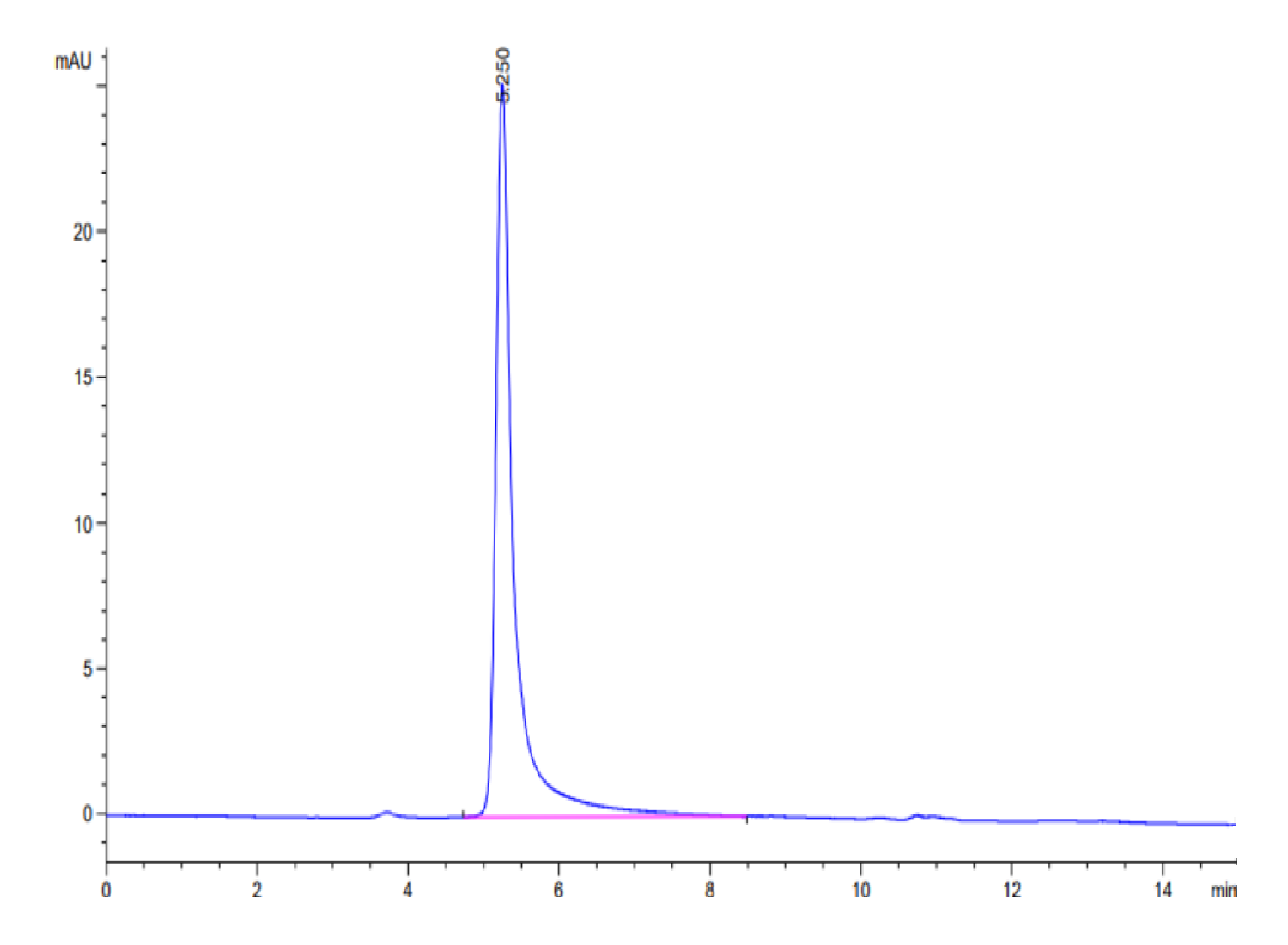
- Purity: > 95% as determined by Bis-Tris PAGE; > 95% as determined by HPLC
- Target: Envelope glycoprotein E
- Species: Varicella-zoster virus (strain Oka vaccine)
- Shipping: Shipped at ambient temperature.
- Synonyms: Envelope glycoprotein E; gE; ORF68;
- Endotoxin: Less than 1 EU per μg by the LAL method.
- Background: VZV glycoprotein E (gE) is most abundantly expressed on the surface of infected cells, and is an essential component for virus replication and cell-to-cell transmission. It is also the main target of virus-specific antibodies and T cell responses that is often selected as vaccine candidate antigen.
- Description: Recombinant Varicella-zoster virus (strain Oka vaccine) Envelope glycoprotein E Protein is expressed from HEK293 with His tag at the C-Terminus.; It contains Ser31-Tyr538.
- Formulation: Lyophilized from 0.22 μm filtered solution in PBS (pH 7.4). Normally 8% trehalose is added as protectant before lyophilization.
- Biotinylated: no
- Exact Sequence: Ser31-Tyr538
- Express System: HEK293
- Geneprotein Id: Q9J3M8
- Reconstitution: Centrifuge the tube before opening. Reconstituting to a concentration more than 100 μg/ml is recommended. Dissolve the lyophilized protein in distilled water.
- Molecular Weight: The protein has a predicted MW of 58.39 kDa. Due to glycosylation, the protein migrates to 65-85 kDa based on Bis-Tris PAGE result.
- Stability and Storage: -20 to -80°C for 12 months as supplied from date of receipt.;-80°C for 3 months after reconstitution.;Recommend to aliquot the protein into smaller quantities for optimal storage. Please minimize freeze-thaw cycles.